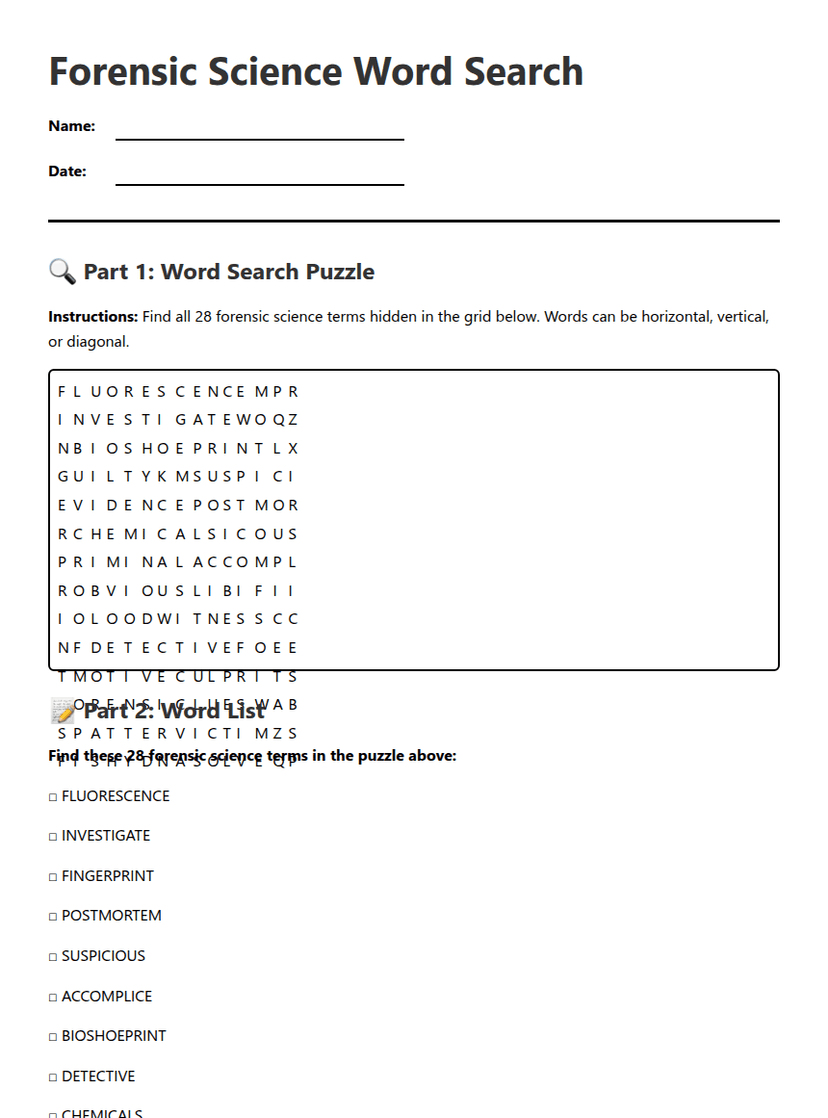
Covalent Bonding Practice Worksheet
A free, printable science worksheet ready for your classroom. Download instantly, print, and hand out to your students — no account needed.

Covalent Bonding Practice Worksheet
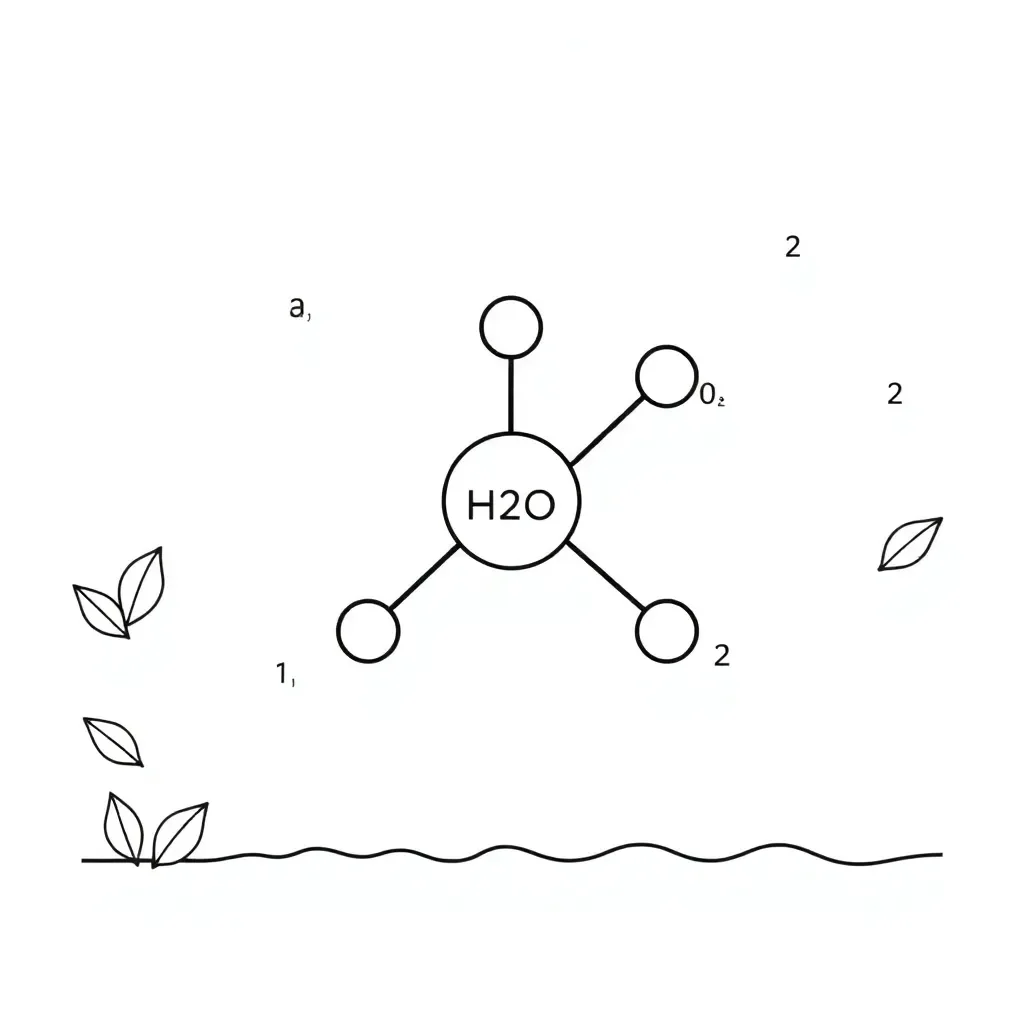
📚 Part 1: Reading Comprehension
What is Covalent Bonding?
Covalent bonding occurs when atoms share electrons to form molecules. Unlike ionic bonding where electrons are transferred, covalent bonds involve atoms sharing one or more pairs of electrons. This sharing allows atoms to achieve a stable electron configuration, similar to the nearest noble gas. Covalent bonds typically form between non-metal atoms and create molecules such as water (H₂O), oxygen gas (O₂), and hydrogen gas (H₂). The shared electrons are attracted to the nuclei of both atoms, holding the atoms together in a strong bond.
🔬 Part 2: Worked Examples
Example 1: Hydrogen Gas (H₂)
Each hydrogen atom has 1 electron and needs 1 more to be stable (like helium). Two hydrogen atoms share their electrons, forming a single covalent bond: H-H
Example 2: Oxygen Gas (O₂)
Each oxygen atom has 6 outer electrons and needs 2 more to be stable (like neon). Two oxygen atoms share 2 pairs of electrons, forming a double covalent bond: O=O
Example 3: Water (H₂O)
Oxygen needs 2 electrons, and each hydrogen needs 1. Oxygen shares one electron with each hydrogen atom, forming two single covalent bonds: H-O-H
✏️ Part 3: Practice Questions
In covalent bonding, atoms _____________ electrons to achieve a _____________ electron configuration.
About This Worksheet
Free Download
No sign-up, no email, no paywall. Just download and print.
Print-Ready
Formatted for standard paper. Clean layout, easy to read.
AI-Generated
Created with Kuraplan's AI, designed for real classroom use.
For Teachers & Parents
Use in classrooms, for homework, tutoring, or homeschool.
Need a custom version of this worksheet?
Kuraplan's AI generates custom worksheets in seconds — differentiated for every learner, aligned to your curriculum.
Generate Custom Worksheets — Free